a bit of info 
Fool's gold may prove an unlikely alternative to overexploited catalytic materials


Fool's gold may prove an unlikely alternative to overexploited catalytic materials
Catalytic materials, which lower the energy barriers for chemical reactions, are used in everything from the commercial production of chemicals to catalytic converters in car engines. However, with current catalytic materials becoming increasingly expensive, scientists are exploring viable alternatives.
Researchers at the University of Cambridge have now discovered that the sulphide material iron pyrite, commonly known as 'Fool's Gold', may hold the answer. Their findings were published online today, 10 February, in Physical Chemistry Chemical Physics.
In the past, sulphur was believed to be one of the most detrimental elements for surface chemical reactions, able to decrease dramatically the reactivity of a catalyst by occupying (poisoning) the "active sites" on the material, but more recently some sulphur materials (for example, molybdenum sulphides) have actually shown interesting catalytic properties of their own.
Using state-of-the-art electronic structure calculations, researchers led by Stephen Jenkins at the University's Department of Chemistry, explored the potential catalytic activity of iron pyrite, the most abundant sulphur mineral on Earth. In their study, the Cambridge researchers focused on the reactions between iron pyrite and nitrogen oxides (NOx), an extremely poisonous class of compounds produced (among other sources) by car engines and industrial power plants.
In the past, sulphur was believed to be one of the most detrimental elements for surface chemical reactions, able to decrease dramatically the reactivity of a catalyst by occupying (poisoning) the "active sites" on the material, but more recently some sulphur materials (for example, molybdenum sulphides) have actually shown interesting catalytic properties of their own.
Using state-of-the-art electronic structure calculations, researchers led by Stephen Jenkins at the University's Department of Chemistry, explored the potential catalytic activity of iron pyrite, the most abundant sulphur mineral on Earth. In their study, the Cambridge researchers focused on the reactions between iron pyrite and nitrogen oxides (NOx), an extremely poisonous class of compounds produced (among other sources) by car engines and industrial power plants.









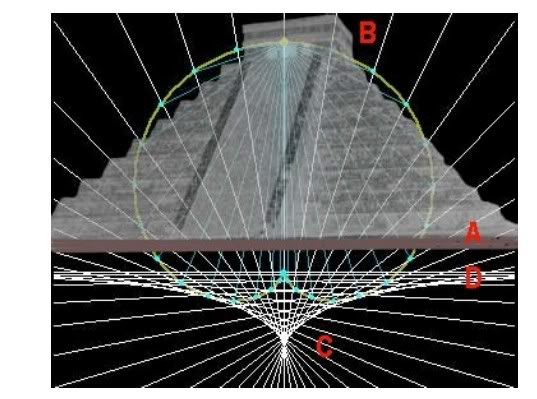
 ...here are a few posts to add to the properties of pyramid and help you see clearer
...here are a few posts to add to the properties of pyramid and help you see clearer 















 )....we would not be this advanced in our discussion if it was not for you guys...so you deserve much respect for that and the blessings of Angels .....thank you guys
)....we would not be this advanced in our discussion if it was not for you guys...so you deserve much respect for that and the blessings of Angels .....thank you guys 

Comment